Pushing Beyond the Limits of Today's CAR T Therapies
Successful CAR T therapy means remission that lasts. To get there, CAR T cells must expand, persist, and stay powerful — even without toxic pre‑conditioning chemotherapy. They need to stay active in the tumor microenvironment, target cancer from multiple angles, and resist immune rejection. iPSC‑derived CAR T cells can be engineered to do exactly that — built for durability and long‑term tumor control.

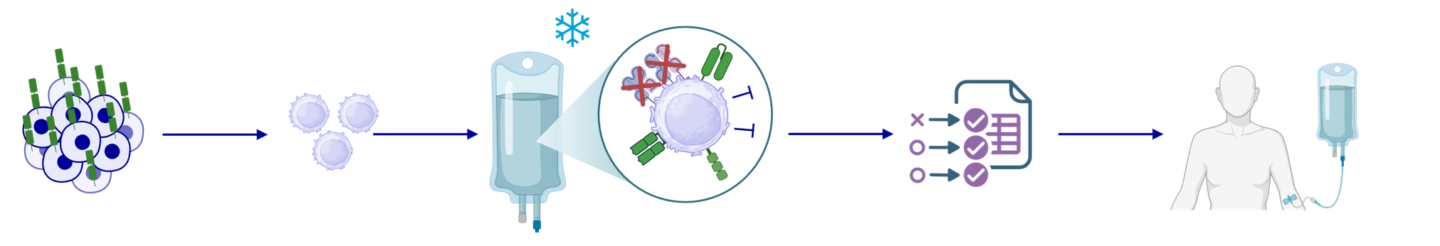
Manufacturing iPSC-CAR T Cells
GMP‑grade iPSCs are genetically engineered and differentiated into iPSC‑derived CAR T cells, which are subsequently expanded, cryopreserved, and subjected to comprehensive quality control. Their off‑the‑shelf availability and suitability for outpatient administration substantially enhance therapeutic accessibility.
Our proprietary manufacturing process delivers batch sizes on par with donor‑derived CAR T cells and is designed for seamless upscaling. With projected production costs below €2,000 per dose, it offers a highly competitive alternative to in vivo CAR T approaches.
CAR iγδ T Cells - A Powerful Cell Therapy Modality
At Evotec, we are developing a versatile iPSC-derived CAR γδ T cells (CAR iγδ T) platform designed to use as cell therapy modality in both oncology and immunology.
γδ T cells are particularly well suited for cell therapy because they:

ICE: Immune cell engager.
TME: Tumor microenvironment.
- Pose a lower risk of graft‑versus‑host disease thanks to their MHC‑independent antigen recognition.
- Reduce the likelihood of tumor escape by recognizing multiple tumor antigens through their natural receptors.
- Naturally infiltrate solid tumors with high efficiency.
- Have demonstrated clinical efficacy with a favorable safety profile and minimal adverse events.
Evotec's Immune-Shieled iγδ T Cell Platform
Evotec’s GMP-compatible gene editing process enables a versatile iγδ T cell platform:

- Depletion of MHC surface display to prevent allorejection via T cells
- Inhibitory mechanism to prevent allorejection via NK cells
- Customizable (exchangeable) CAR for target specificity or combination with Immune Cell Engager (ICE)
- Safety switch for therapeutic intervention
- γδTCR to recognize natural tumor antigens
- Gene edits(s) for persistency and TME-resistance
Evotec’s CAR iγδ T cells to treat liver cancer
Evotec is currently developing an immune-shielded CAR iγδ T cell product candidate against liver cancer.
Competitiveness of CAR iγδ T Cells

Learn More About Our Science in Our Articles & Posters
- Genetic Integrity Testing of Genome-Edited Pluripotent Stem Cell Lines Used for Cell Therapy Applications >
- Powering the Next Generation of Cancer Immunotherapy: iPSC-Derived Immune Cells Meet Immune Cell Engagers >
- From Bench to GMP: Streamlining the Transfer of Cell-Based Processes to Clinical Grade Manufacturing at Evotec’s Manufacturing Site >
- Off the Shelf Immune-Shielded Stem Cell-Derived γδ T Cells for Clinical Applications >
