Abstract
High performance cell culture media provide an essential environment to ensure cells are producing high titers and high quality biotherapeutics. These attributes are important drivers in determining whether a biotherapeutic can be manufactured commercially with economics that support market launch. In this report we describe the use of a proprietary chemically-defined (CD), animal origin free (AOF) Chinese Hamster Ovary (CHO) perfusion cell culture medium designed to maximize productivity and cell viability across scales, while maintaining critical product quality attributes, which is marketed by Just – Evotec Biologics under the J.Media™ trademark. The proprietary formulation is room temperature stable and able to be concentrated 5× making it attractive for clinical and commercial use at both small and large scale. J.Media™ perfusion medium performance was compared to commercially available perfusion media using two IgG1-expressing CHO cell lines in small scale mock perfusion study, and performance scalability was tested from 3 L bench scale to 500 L single use bioreactors (SUB). Results show market-leading volumetric and specific productivity by efficiently funneling nutrients towards protein production while maintaining consistent critical quality attributes (CQAs) including glycans such as high mannose, sialylation, β-galactose, and afucosylation.
Introduction
CHO cell culture is the production system of choice for most monoclonal antibodies (mAbs) due to CHO cells’ ability to provide relatively high productivity and preserve glycosylation patterns similar to those found in humans1, thereby supporting the desired clinical PK/PD and reducing risk of deleterious attributes such as immunogenicity. As of early 2025, there are over 200 therapeutic mAbs on the market with another 1,400 investigational product candidates in development2, a large fraction of which are produced using CHO cell culture.
Over the past decade, the biotherapeutics manufacturing industry has shifted towards using intensified perfusion cell culture over fed batch cell culture formats. This is largely because the intensified perfusion allows for significantly higher cell densities and the delivery of higher product mass reducing the overall cost of goods (COGs). Achieving these benefits requires advanced media formulations capable of supporting high cell densities (often >80x106 cells/mL) while maintaining consistent product quality. To address this need, Just – Evotec Biologics developed a chemically defined, animal origin–free medium optimized for intensified perfusion processes (J.Media™).
In this work, we compare the performance of the J.Media™ proprietary perfusion medium to that of different commercial perfusion cell culture media in a high-throughput, plate-based system that mimics continuous perfusion using two cell lines expressing an IgG1 mAb. The performance of cell culture media was evaluated using key metrics such as volumetric productivity (Vp, g/L/d), specific productivity (qp, pg/cell/d), maximum viable cell density (VCD), and cell viability. Additionally, we evaluate the performance of J.Media™ medium at a 3 L (lab scale) and 500 L (clinical and commercial scale) stirred tank reactors. We compared cell culture performance parameters as well as the levels of potential glycan CQAs such as total high mannose, β-galactose, sialylation, and afucosylation. The results demonstrate that the J.Media™ proprietary perfusion medium drives higher volumetric and specific productivity compared to commercial perfusion media in the tested cell lines and shows robust performance from 3 L to 500 L by maintaining protein product CQAs.
Materials and Methods
Cell Lines and Media
CHO-GS cell lines (J.CHO®) expressing two different IgG1 monoclonal antibodies (mAb1 and mAb2) were chosen to evaluate commercial perfusion media and J.Media™ proprietary perfusion medium performance. These cell lines are growth-factor independent and do not require insulin or insulin-growth factor supplementation in the J.Media™ perfusion medium.
Mock Perfusion cultures
A small-scale mock perfusion (MP) model operating at 2 mL working volume in 24 deep well plates was used for evaluating different perfusion media. Because dissolved gas and culture pH are not actively controlled (as in traditional bioreactors at scale) they are a more stringent test of media performance and robustness. The two cell lines expressing mAb1 and mAb2 were tested in J.Media™ medium and three other commercial perfusion media (Medium A-C) in triplicate. Each well was inoculated at 10 million cells/mL, and plates were incubated in Kuhner shakers maintained at 37°C, 5% CO2, 220 rpm and orbital throw of 50 mm. Once per 24 hours, cells were pelleted by centrifugation, and 80% of the spent culture medium was removed and replaced with fresh medium (0.8 vessel volumes, or 0.8 VVD, exchanged) to mimic perfusion cell culture. Growth and cell viability were monitored daily using a CellacaMX cell counter. Bleeding of the culture to maintain a target cell density was not performed. Glucose, lactate, ammonia, and IgG titer levels were measured in spent media samples using a Roche CEDEX Bio HT instrument. Glucose was supplemented as required to maintain >2 g/L in the culture.
3 L and 500 L Bioreactor Perfusion Culture
A 3 L glass bioreactor (1.6 L working volume) was used as a bench-scale model of a 500 L single use bioreactor (SUB) manufacturing scale (450 L working volume). Operating parameters were scaled from 3 L scale to 500 L scale to account for differences in equipment between the scales. Cells were seeded at 0.6 million cells/mL in commercially available expansion media (CDOptiCHO with 5 g/kg poloxamer). Both bioreactor scales were equipped with a recirculation loop containing a 0.2 μm perfusion filter to allow cell retention and continuous media exchange, while allowing the product to pass through into the permeate. The perfusion of fresh J.Media™ perfusion medium was initiated on day 2 and the perfusion rate gradually increased to 2 vessel volumes per day (VVD) by day 5 of the 15-day process. A target density of 80 million cells/mL was maintained with an automated culture bleed control loop that used either a daily ViCell cell count and bleed calculation (500 L scale), or a capacitance probe with a set permittivity target (3 L scale) determined for each clonal line. The pH was controlled to target using CO2 and sodium carbonate. The dissolved oxygen (DO) target was maintained by sparging using air and O2 control loops. Glucose was monitored daily using a Nova Flex2 and was maintained at sufficient levels by a concentrated feed. Antifoam was added as needed to control foam. Permeate was directed to waste until day 7 followed by permeate harvest from day 7 (D7) to day 15 (D15). Permeate samples from 3 L and 500 L bioreactors were taken on various days between day 8 and 14 and purified by Protein A chromatography prior to analysis. The data shown for the 3 L and 500 L bioreactor runs represents data from summarized from one or more bioreactor runs.
Analytical Assays
Permeate protein titer was measured using a ultra-high performance liquid chromatography (UHPLC)-based Protein A chromatography method. The bound protein was eluted under acidic conditions and the area under the peak at 280 nm was calculated and quantitated against a six-point standard curve.
Size exclusion ultra-high performance liquid chromatography (SE-UHPLC) was used to quantitate high molecular weight (HMW) species. Purity and HMW levels were determined by % Peak Area using UV absorbance at 220 nm.
Reduced capillary electrophoresis (rCE-SDS) was used to assess purity of a molecule by resolving low molecular weight (LMW) and medium molecular weight (MMW) species after treatment with the reducing agent β-mercaptoethanol. LMW and MMW species were determined by calculating % Time Corrected Peak Area for the peaks detected by UV absorbance at 220 nm.
A hydrophilic interaction liquid chromatography (HILIC) glycan mapping method was used to characterize Fc N-linked glycans for mAb2. Glycans were released from the protein via PNGase F digestion and labeled with procainamide. Glycans were then separated by HILIC in-line with a fluorescence detector and identified based on their retention time compared to previously analyzed standards.
A multi-attribute-method (MAM) using reversed phase LC-MS/MS with detection by a Thermo Scientific Q Exactive™ HF mass spectrometer was used for characterization glycosylation of mAb1 that has two glycosylation sites. MAM provided quantification of glycan related CQAs for both the Fc-linked glycan site (N311) and the Fab linked glycan site (N76).
Results
Small scale mock perfusion evaluation
A small-scale MP model was used to rapidly screen and benchmark J.Media™ culture medium against three commercial media formulations (Medium A–C) and filter out formulations that do not support growth, as was the case for medium C with the mAb1-expressing cell line (data not shown). The performance was quantified in an 8-day experiment through evaluation of metrics known to be important in the manufacture of biotherapeutics including: maximum supported VCD, Vp, qp, and process duration, which is dependent on maintaining cell viability (Figure 1). J.Media™ medium showed the highest specific productivity (100–120 pg/cell/d, Figure 1 A). At day 8, mAb1 productivity with J.Media™ medium was ~200% higher than medium B and comparable to medium A, whereas mAb2 production was ~200% higher than media B and C and 14% higher than medium A. Enhanced qp supported by J.Media™ medium drives the high volumetric productivity (Vp) of 3–4 g/L/d for both mAb1 and mAb2 (Figure 1 B). For mAb1, this is 26% higher than medium A, and over 200% higher than medium B. For mAb2, this is over 200% higher than medium C.
In addition to supporting cells to deliver high volumetric and specific productivities, it is essential that the media can sustain high cell densities and viability over the culture duration. While VCD values attained in the plate-based MP model are not directly translatable to maximum VCD achievable in stirred-tank reactors, (where dissolved gas and culture pH are actively controlled) trends in MP VCD are generally predictive of stirred-tank reactor cell density. In this experiment, J.Media™ medium maintained 30–40 million cells/mL VCD with both mAb1 and mAb2 cell lines (Figure 1 C). This was higher than medium A, and comparable to medium B for mAb1, while it was higher than medium C, and comparable to medium A and B for mAb2. Maintaining high viability is crucial to extended perfusion processes, and we find that J.Media™ medium can maintain high viability (>90%) over the course of this 8-day MP (Figure 1 D).
Finally, the cumulative titer from the 8-day MP process was evaluated (Figure 1 E). J.Media™ medium showed statistically higher cumulative titers for both mAb1 and mAb2 when compared to the media A–C as identified using Tukey’s HSD test, due to the robust cell culture performance metrics demonstrated in Figure 1 A–D. The cumulative titer data highlight the advantages of J.Media™ medium, which enables large product yield.
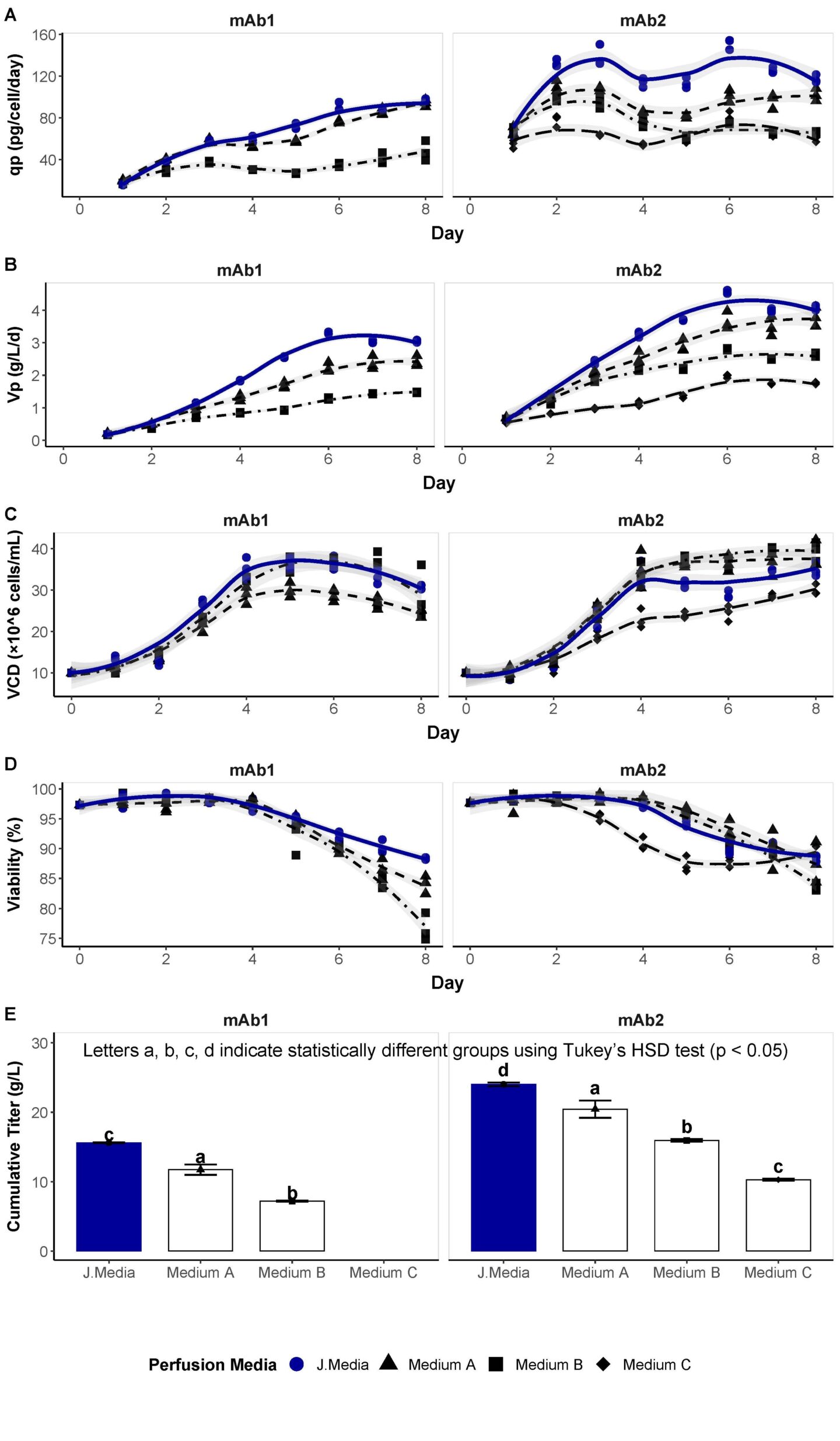
Figure 1. Mock Perfusion cell culture parameters.
CHO-GS cells expressing mAb1 and mAb2 were grown in 24 DWP at working volume 2 mL. (A–D) Blue line, circle: J.Media™ medium, black line, triangle: Medium A, black line, square: Medium B, black line, diamond: Medium C. Shaded region shows the confidence interval (α = 0.05). A) Specific productivity (qp), (B) Volumetric productivity (Vp), (C) Viable cell density (VCD), (D) Viability (%), and (E) Cumulative titer data is shown for 3 technical replicates each for J.Media™ medium and three commercial perfusion media, where letters indicate statistically different groups based on Tukey’s HSD test. Blue bar: J.Media™ medium; white bars: commercial media A. B, and C.
We also calculated cell metabolic parameters such as cell-specific exchange rates for glucose uptake, and lactate and ammonia secretion (Figure 2). In general, during the first four days of rapid growth, exchange rates for these metabolites were higher compared to the last four days of culture when cells reach a steady VCD. The specific glucose consumption rate was similar for all conditions (Figure 2 A), but key differences were observed in cell-specific rate of exchange of waste products, such as lactate (Figure 2 B) and ammonia (Figure 2 C) that can be detrimental to cell growth and viability3. Lactate secretion diverts carbon that could be used to produce biomass or IgG, and its accumulation can be detrimental to cell
growth. CHO cells often convert >60% of glucose into lactate4, indicating that reducing its secretion is a key target to improve protein yield from media nutrients. J.Media™ medium consistently showed the lowest apparent lactate secretion rates in both mAb1 and mAb2 in all stages of the experiment, indicating that J.Media™ culture medium promotes efficient
nutrient utilization. This observation was supported by the high qp previously described (Figure 1 A). J.Media™ medium also showed moderate ammonia secretion, below levels observed in media B and C for both mAb1 and mAb2.

Figure 2. Mock Perfusion cell metabolism parameters.
CHO-GS cells expressing mAb1 and mAb2 were grown in 24 DWP at working volume 2 mL. Spent media was sampled daily to measure glucose, lactate, and ammonia. (A) Specific glucose uptake rate, (B) specific lactate secretion, and (C) specific ammonia secretion rate. Data are shown for 3 replicates of J.Media™ medium and three commercial perfusion media. (A-C) Blue line, circle: J.Media™ medium, black line, triangle: Medium A, black line, square: Medium B, black line, diamond: Medium C. Shaded region shows the confidence interval (α = 0.05).
Bioreactor performance and scalability at 3 L and 500 L
Gathering data at multiple scales, from bench to manufacturing scale, allows comparison across scales and evaluation of the representativeness of model systems. Although MP models provide higher throughput for screening investigations, these models differ from stirred tank bioreactor perfusion processes in several key elements. Unlike MP, bioreactors can maintain pH and DO setpoints, offer increased agitation and continuous perfusion of media, as well as continuously control cell density to a target setpoint via on demand cell bleeding of the bioreactor based on in-line capacitance monitoring. These control parameters can result in differences in steady state VCD, as well key performance metrics such as Vp.
To evaluate J.Media™ medium in stirred tank bioreactors, the mAb1 and mAb2 cell lines were grown in 3 L bioreactors and a 500 L single use bioreactor (SUB) and monitored for VCD, Vp, viability, and qp (Figure 3). Compared to the MP model, the 3 L bioreactors could maintain a ~2.5-fold higher VCD, likely due to control of pH and DO, and increased gas exchange and continuous perfusion at a higher VVD. The MP model was able to predict the rank order of productivity of the 3 L model, where mAb2 had greater volumetric productivity than mAb1.
To facilitate the scalability evaluation, we looked at replicate runs listed in Table 1.
The 3 L and 500 L VCD data show good agreement despite the difference in culture density control (capacitance target for 3 L vs VCD target using ViCell XR for 500 L, Figure 3 A). There is also agreement between the models in terms of Vp, qp (Figure 3 B, D) and viability, although the 500 L runs show slightly higher viability compared to 3 L bioreactors for mAb1 and slightly lower ending viability in the 500 L for mAb2 (Figure 3 C).
Finally, J.Media™ medium enabled high perfusion process productivity for mAb1 and mAb2 in a 15-day process. For example, a single 500 SUB (450 L working volume), was able to yield 9.14 kg mAb1 and 12.56 kg mAb2. This yield, in a traditional fed batch setup, requires 5–10 fold larger bioreactors scales considering an average fed-batch titer of 3.2 g/L5, or several runs, thus highlighting the advantages of J.Media™ medium in perfusion processes.

Figure 3. Evaluation of key cell culture parameters in J.Media™ medium on scale up for mAb1 and mAb2.
(A) VCD (million cells), (B) Vp, (g/L/d), (C) Viability (%), (D) qp (pg/cell/day), and (E) Cumulative Permeate Titer (g/L) from day 7 to day 15. The number of replicate runs are: 3 L mAb1 = 5, 500 L mAb1 = 2, 3 L mAb2 = 3, 500 L mAb2 = 3. (A-D) Grey line, circle: 3 L, Blue line, circle: 500 L. (E) Grey bars: 3 L, Blue bars: 500 L.
Evaluation of mAb product quality upon scale up in J.Media™ medium
We next evaluated the impact of scale up in J.Media™ medium from 3 L to 500 L on mAb quality. We utilized SEC to quantify mAb aggregation and rCE-SDS to quantify LMW and MMW species that indicate clipping and fragmentation (Figure 4). To determine the net product quality, we focused on days 7 to 15 when product-containing permeate is typically harvested continuously for downstream processing. The CQA attribute levels of the proteins produced change over the course of the collection period, and the total amount of the CQA is that of the pooled product at the end of the process. The net product quality (%) for these attributes is calculated as:
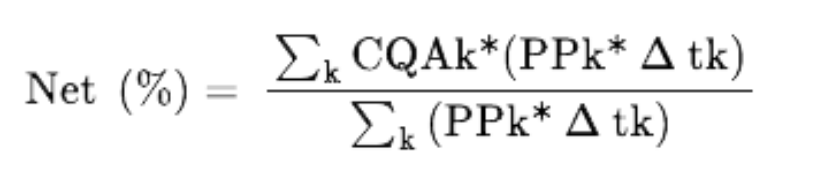
Here, PPk is permeate productivity at time k, CQAk is the critical quality attribute % measured at time k, and Dtk is the time interval. This was done only where we had CQA data available, which was typically day 8, 10, 12, and 14.
It is essential that the perfusion medium can main-tain product quality attributes when scaled up, and we find that there is no significant impact on these characteristics in either mAb1 or mAb2 upon scale up in J.Media™ medium.


Figure 4. Evaluation of product quality parameters in J.Media™ medium on scale up for mAb1 and mAb2.
Data represent mean and error bars showing standard deviation of a different number of replicates. The number of replicate runs are: 3 L mAb1 = 5, 500 L mAb1 = 1, 3 L mAb2 = 4, 500 L mAb2 = 2. Grey bars: 3 L, Blue bars: 500 L.
Evaluation of mAb glycoslation upon scale up in J.Media™ medium
A crucial aspect in process development of biotherapeutics is maintaining consistent levels of glycan species during scale up. Figures 5 and 6 present the levels of N-linked glycan species of mAb1 and mAb2 at 3 L and 500 L scale. The single Fc N-linked glycan site on mAb2 was characterized using the HILIC method (Figure 5), while for mAb1, which contains both an Fc and a Fab N-linked glycosylation site, the glycans were characterized using MAM so that the glycan profile of each site could be determined (Figure 6).

Figure 5. Evaluation of Fc N-linked and Fab N-linked glycan site in J.Media™ medium at 3 L and 500 L scale for mAb1 using MAM method. 3 L and 500 L N = 1. Grey bars: 3 L, Blue bars: 500 L.
J.Media™ medium was able to support consistent growth and product quality as demonstrated by glycan attribute levels with no statistical differences upon scale up from 3 L to 500 L for mAb2.

Figure 6. Evaluation of Fc N-linked and Fab N-linked glycan site in J.Media™ medium at 3 L and 500 L scale for mAb1 using MAM method. 3 L and 500 L N = 1. Grey bars: 3 L, Blue bars: 500 L.
Figure 6 shows that the glycan species for the Fc N-linked and Fab N-linked glycans of mAb1 are roughly comparable between the two scales, with afucosylation being somewhat higher at the 500 L scale for the Fc N-linked glycan whereas the levels of sialylation at 500 L being slightly higher and beta galactose slightly lower for the Fab N-linked glycan.
In summary, our data show that J.Media™ medium is a robust CHO cell culture medium that can facilitate scaling from small-scale process development activities into large-scale operations while maintaining the product quality and glycan attributes that are critical to success of biotherapeutic
efficacy.
Evaluation perfusion media attributes
Selection of perfusion media at scale often factors in several aspects beyond cell culture performance and product quality including storage conditions, expiration time, and concentration capability to reduce storage footprint. Table 2 below shows a comparison of J.Media™ medium with commercial media A, B, and C.
J.Media™ medium is room temperature stable (data not shown) which reduces the requirement of cold storage space, cold chain supply and transport issues as well as extends shelf life. Compared with competitor media we tested, J.Media™ medium offers improved operational flexibility. Perfusion format production systems generally require large volumes of media. J.Media™ medium can be concentrated and prepared as a 5× solution (requires additional feed) and is supplied with WFI directly into the perfusion bioreactor. This also makes the
addition of additional feeds or other components easy if desired. This results in significant reduction in media volume requirements. For example, a 500 L reactor operated for 15 days at 2 VVD requires 3000 L of 5× media concentrate as compared to 15,000 L of a 1× perfusion media.
Conclusion
In this work, we demonstrate that J.Media™ medium can support the desired cell growth, productivity, and quality of CHO cells to produce best-in-class protein production for two IgG1 molecules tested. This is most likely due to a reduction in waste products like lactate and ammonia. However, further assessments of the detailed metabolomic response driven by J.Media™ medium are planned for future studies.
We also demonstrated scalability to clinical/commercial levels using J.Media™ medium. Notably, 500 L scale bioreactor runs in this study utilized a 5× concentrate with feeds shelf stable at room temperature. These two attributes – the ability of the proprietary J.Media™ medium to concentrate to 5× and its long term stability at room temperature – are important for commercial manufacturing considerations such as cold chain storage, transport and shelf life. Both perfusion formats and large scale fed batch (>5000 L) have large media requirements and as such J.Media™ medium use is also highly desirable from a facility footprint utilization viewpoint. Future studies of J.Media™ medium are aimed at increasing shelf life, comparison with commercial media in perfusion bioreactors, as well as the evaluation of performance in commercial CHO cell lines used in both biotherapeutic development and academic research.
References
- Liang, K.; Luo, H.; Li, Q. Enhancing and Stabilizing Monoclonal Antibody Production by Chinese Hamster Ovary (CHO) Cells with Optimized Perfusion Culture Strategies. Front. Bioeng. Biotechnol. 2023, 11, 1112349. https://doi.org/10.3389/fbioe.2023.1112349.
- Crescioli, S.; Kaplon, H.; Wang, L.; Visweswaraiah, J.; Kapoor, V.; Reichert, J. M. Antibodies to Watch
in 2025. mAbs 2025, 17 (1), 2443538. https://doi.org/10.1080/19420862.2024.2443538. - Pereira, S.; Kildegaard, H. F.; Andersen, M. R. Impact of CHO Metabolism on Cell Growth and Protein Production: An Overview of Toxic and Inhibiting Metabolites and Nutrients. Biotechnol. J. 2018,13 (3), 1700499. https://doi.org/10.1002/biot.201700499.
- Ahn, W. S.; Antoniewicz, M. R. Metabolic Flux Analysis of CHO Cells at Growth and Non-Growth Phases Using Isotopic Tracers and Mass Spectrometry. Metab. Eng. 2011, 13 (5), 598–609. https://doi.org/10.1016/j.ymben.2011.07.002.
- Langer, E.; Rader, R. Biopharmaceutical Manufacturing: Historical and Future Trends in Titers, Yields, and Efficiency in Commercial-Scale Bioprocessing. Bioprocess. J. 2015, 13 (4), 47–54. https://doi.org/10.12665/J134.Langer.