Antibody-drug conjugate (ADC), termed a “biological missile”, is leading a new era of targeted therapy. ADCs link target-specific monoclonal antibodies with cytotoxic drugs to efficiently transport the drug to the target cells, reducing toxicity to healthy cells. Evotec’s antibody-drug conjugate services provide the specialized technology and expertise required for bespoke ADC development.
How do antibody drug conjugates work?
ADCs combine monoclonal antibody specificity with potent cytotoxic payloads to selectively deliver drugs to diseased cells. This is achieved through a three-component structure and a selective mode of action.
Antibody Drug Conjugate Structure
ADCs contain three key components (Figure 1):
- Antibody for antigen recognition and targeted drug delivery
- Linker for stability and controlled release
- Payload for tumor-specific cell killing
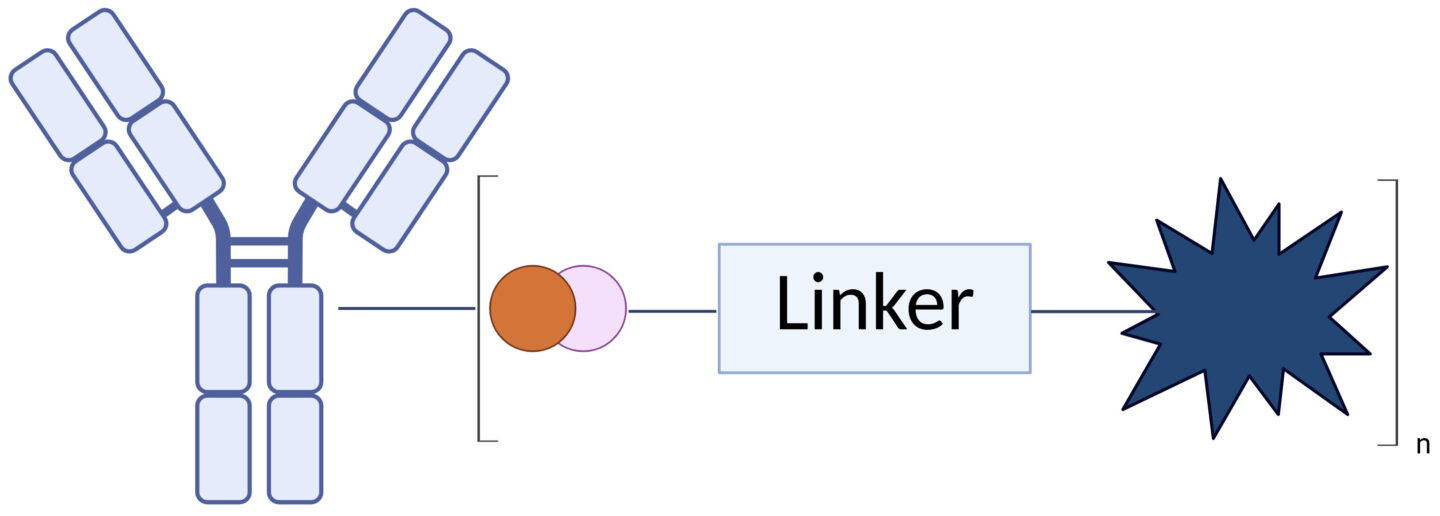
Figure 1. Antibody-drug conjugate structure
Antibody Drug Conjugate Mode of Action
ADCs, delivered via intravenous injection or infusion, seek target cells where antibodies bind to cell-surface antigens. The ADC is then internalized and trafficked via endosomes and lysosomes, where the linker is cleaved or degraded to release the active drug. By only delivering the ADC to target cells, this minimizes systemic toxicity and opens up possible therapeutic applications.
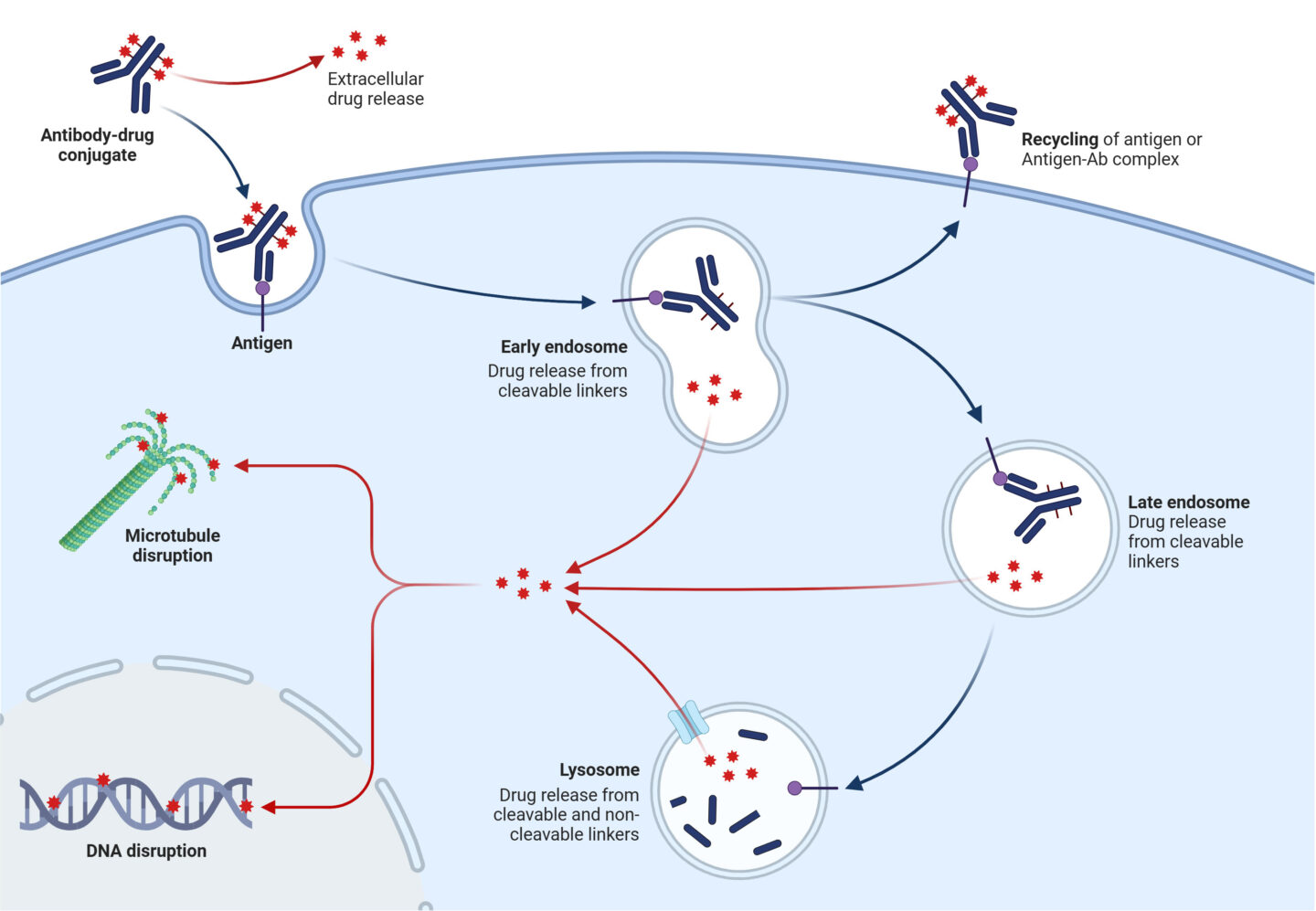
Figure 2. Antibody-drug conjugates: mode of action
In chemotherapy, ADCs combine high specificity with potent killing to accurately target cancer cells while minimizing toxicity to healthy cells. Beyond established cytotoxics and oncology, researchers are investigating ADCs as therapeutic candidates for challenging indications such as autoimmune diseases and persistent bacterial infections.
Evotec’s Comprehensive Antibody Drug Conjugate Services
Evotec delivers end-to-end antibody drug conjugate services from gene to IND, with flexible entry points throughout the preclinical discovery process (Figure 3). Our integrated platform combines disease expertise with the advanced technologies required to accelerate ADC development.
Antibody Drug Conjugate Development Process: Target to IND
Target Identification & Validation
Target identification pinpoints specific biological molecules that drive disease progression and can be modulated therapeutically. Our approach includes:
- Bioinformatics database screening to identify prevalent target expression in tumors
- Homogeneity and expression level analysis in tumor versus normal tissues
- Shedding assessment to measure target stability
- Target internalization validation for optimal ADC uptake
Antibody Discovery (5-7 months)
High-affinity antibody hit identification establishes the foundation for effective ADCs. We select internalizing antibodies with optimal binding profiles, then identify up to 10 monoclonal antibodies that best fit the intended linker and payload strategy.
ADC Lead Generation & Selection (6 months)
Linker, payload, and drug-to-antibody ratio (DAR) optimization creates ADC candidates with controlled release profiles. Our medicinal chemistry expertise assesses stability while maximizing therapeutic impact and minimizing off-target toxicity.
ADC Candidate Profiling (6 months)
Full characterization of ADC candidates evaluates 1-3 lead molecules across multiple parameters. This rigorous profiling identifies the optimal candidate for preclinical development and regulatory submission.
ADC Preclinical Development (9-12 months)
Regulatory studies and IND-enabling work prepare candidates for clinical trials. Parallel antibody CMC (Chemistry, Manufacturing, Controls) development and payload/ADC CMC development assess manufacturing readiness.
Integration Throughout Development
Disease-Relevant Models
In vivo and in vitro studies use primary cells and iPSC-derived disease models that authentically replicate human biology. These systems preserve native disease mechanisms, cellular complexity, and patient-specific genetic backgrounds, making validation results more predictive of clinical outcomes.
Rigorous Toxicology Assessments
Cyprotex’s capabilities identify compounds with optimal safety profiles for clinical advancement. Since drug toxicity drives significant attrition, early toxicity identification and mechanism understanding save time and development costs while progressing the safest compounds.
Patient Sample Access
Access to patient samples provides clinically relevant validation throughout development, bridging preclinical findings to human disease biology.
This comprehensive process develops high-quality ADCs with demonstrated potential for clinical success, typically advancing from target nomination to IND in 26-31 months, depending on entry point and program complexity.

Figure 3. Integrated antibody drug conjugate development from target validation to IND
Advanced Antibody Drug Conjugate Optimization Capabilities
Through integrated antibody discovery, medicinal chemistry, linker technology, and payload engineering, Evotec optimizes ADCs for stability, controlled release, and superior therapeutic profiles. Our approach delivers:
- Antibody engineering for optimal antigen binding and internalization
- Medicinal chemistry for potent payload development
- Linker technology engineered for stability and controlled release
- Payload engineering to maximize therapeutic impact
- Reduced systemic toxicity, increasing therapeutic applications
Infrastructure
Our ADC platform leverages the entire Evotec/ Cyprotex multi-modality infrastructure for bespoke studies. We work with all types of drug molecules, from small to large. This approach addresses different aspects of disease biology simultaneously.
Partnership Model
Evotec has collaborative partnerships with pharmaceutical and biotechnology companies, as well as academic institutions. These partnerships promote efficient identification and optimization of novel ADC candidates, accelerating the creation of next-generation therapies.
Partnering for Antibody Drug Conjugate Success
Evotec brings integrated gene-to-IND antibody-drug conjugate capabilities to strategic partnerships.
Our approach combines target validation, antibody discovery, conjugation chemistry, and preclinical development, accelerating timelines to 26-31 months.
Access to patient samples and iPSC-derived disease models boost clinical translatability, while Cyprotex's toxicology capabilities derisk programs early when it's most cost-effective.
Our multi-modality infrastructure addresses disease biology from multiple angles, creating robust therapeutic candidates.
Flexible entry points accommodate your program at any stage, from target identification through IND submission.
Whether you're a pharmaceutical partner, biotechnology innovator, or academic institution, Evotec's ADC expertise and integrated facilities accelerate next-generation therapies.
Ready to advance your antibody drug conjugate program? Contact our team to discuss how our integrated platform can transform your concept into a clinical candidate.
Frequently Asked Questions
What are antibody drug conjugates (ADCs)?
ADCs selectively deliver therapeutic payloads to disease cells while sparing healthy tissues. They contain three components: an antibody for antigen recognition, a linker for controlled release, and a payload for targeted cell killing. This targeted approach minimizes systemic toxicity while maximizing therapeutic impact.
How long does ADC development take from target to IND?
Evotec's integrated platform accelerates development to 26-31 months, depending on entry point and program complexity. Our gene-to-IND pathway includes antibody discovery (5-7 months), lead optimization (6 months), candidate profiling (6 months), and preclinical development (9-12 months). Multiple entry points allow partners to engage at any stage.
What types of payloads can be conjugated to antibodies?
Beyond traditional cytotoxics for oncology, Evotec's conjugation strategies explore diverse payloads including protein degraders, antibiotics, immune modulators, peptides, and oligonucleotides. Our medicinal chemistry expertise optimizes payload selection and linker technology for stability and controlled release. This versatility expands ADC applications beyond cancer to autoimmune diseases and bacterial infections.
How do you assess ADC safety & reduce toxicity?
Rigorous toxicology assessments identify compounds with optimal safety profiles before clinical advancement. Our Cyprotex capabilities detect potential toxicity early, understanding mechanisms to save time and costs. Disease-relevant models using primary cells and iPSC-derived systems make validation results more predictive of clinical outcomes, while targeted delivery minimizes off-target effects.