GPCRs at the Center of Modern Medicine
G protein–coupled receptors (GPCRs) are one of the largest and most diverse families of drug targets. They sit at the cell surface, acting as molecular sentinels that detect signals from the external environment and translate them into cellular responses. This ability makes them central to processes as varied as cardiovascular regulation, immune defense, and neurological signaling.
For decades, GPCRs have been a cornerstone of drug discovery, with many approved therapies targeting them. Yet despite their prominence, GPCR drug development remains challenging. The family is vast, with receptors that share structural similarities but differ in subtle ways. This overlap complicates selective targeting. Moreover, GPCRs are involved in multiple biological pathways, making it difficult to link a specific receptor to a single disease outcome.
The result is high attrition: many GPCR programs fail in clinical development. Not because the targets are irrelevant, but because the industry lacks robust tools to measure whether a drug is working in the right patient, at the right time. Biomarkers are increasingly recognized as the solution to this problem.
Biomarkers as the Missing Link
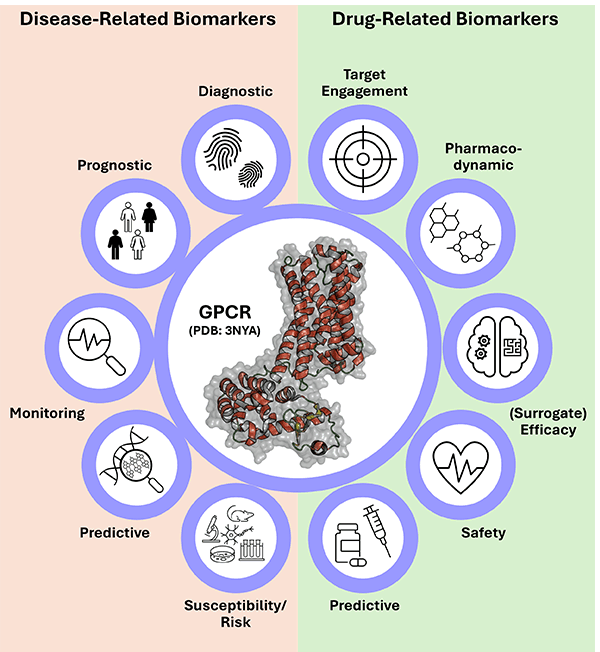
Biomarkers are measurable indicators of biological states or responses, such as proteins, genes, metabolites, imaging signals, or physiological readouts. In drug development, they serve as the missing link between preclinical promise and clinical success.
Source: https://doi.org/10.1016/B978-0-443-29808-0.00036-4
There are biomarkers that are related to disease or a condition:
- Diagnostic biomarkers identify patients with a specific disease subtype.
- Prognostic biomarkers predict how a disease will progress.
- Monitoring biomarkers track disease evolution and treatment impact over time.
- Response biomarkers demonstrate whether a drug engages its target and produces the desired effect.
- Predictive biomarkers: Indicate whether a patient is likely to respond to a specific therapy. They guide patient stratification both in clinical trials and in real-world practice, ensuring that treatments are targeted to those most likely to benefit.
- Susceptibility or risk biomarkers: Highlight an individual’s predisposition to developing a disease or experiencing earlier onset. For example, genetic variants such as APOE alleles in Alzheimer’s disease can signal elevated risk even before symptoms appear.
And there are biomarkers that are related to a drug or a treatment response:
- Target engagement biomarkers: Provide direct evidence that a drug is binding to and modulating its intended receptor or pathway. In GPCR research, receptor binding assays or PET tracers are often used to confirm engagement.
- Pharmacodynamic or surrogate efficacy biomarkers: Demonstrate functional changes downstream of target engagement, offering early signals of therapeutic effect. Examples include shifts in signaling cascades or measurable changes in disease-related proteins.
- Safety response biomarkers: Detect potential adverse effects early, helping developers manage risk. Kidney injury molecule 1 (KIM 1), for instance, has proven useful in identifying renal toxicity before traditional markers show abnormalities.
- Predictive biomarkers: Identify patients most likely to benefit from specific therapies, improving trial success and guiding clinical practice. In inflammatory bowel disease, genetic variations and molecular signatures help exclude non‑responders, ensuring targeted treatment and reducing costly trial failures.
By integrating biomarkers into trial design, companies can reduce uncertainty, streamline patient selection, and increase the likelihood of success. In oncology, biomarker-guided trials have shown nearly double the approval rates compared to those without biomarker integration. This demonstrates their strategic value not only in science but also in business outcomes.
Why So Many Trials Fail
The statistics are sobering: between 2011 and 2020, fewer than 1 in 10 drugs entering Phase 1 reached approval. The leading cause of failure was lack of efficacy. This often reflects four issues:
- Target mis selection: choosing a receptor that is not truly relevant to the disease.
- Suboptimal drug modality: developing a compound that cannot effectively engage the target.
- Inappropriate patient inclusion: enrolling patients unlikely to benefit.
- Insufficient evidence of target engagement: failing to prove the drug is active in the intended tissue.
Biomarkers directly address these challenges. They provide evidence of target engagement, help stratify patients, and allow early detection of efficacy signals. In essence, they transform trial design from guesswork into a data driven process. For pharma and biotech companies, this means fewer costly failures and faster paths to approval.
Case Studies: Biomarkers in GPCR Development
Adenosine A2A receptor:
This receptor has been implicated in cancer progression and Parkinson’s disease. Elevated expression levels can serve as a prognostic marker in oncology, guiding patient selection. In neurology, the FDA approved drug istradefylline (Nourianz®) illustrates both the promise and the challenge. Approval was delayed for years due to the absence of sensitive biomarkers for Parkinson’s progression, highlighting how biomarker gaps can slow innovation.
S1P receptors:
Sphingosine 1 phosphate (S1P) receptors regulate immune cell trafficking. In multiple sclerosis, fingolimod (Gilenya®) works by reducing circulating lymphocytes. Here, a simple blood count serves as a powerful biomarker—simultaneously demonstrating target engagement, pharmacodynamic response, and disease impact. This case exemplifies how a straightforward biomarker can accelerate development and support regulatory approval.
Cannabinoid receptors:
Part of the endocannabinoid system, these receptors are emerging targets in neurology and immunology. Biomarker strategies are helping distinguish therapeutic applications from broader physiological effects, paving the way for precision interventions in conditions such as epilepsy, pain, and autoimmune disorders.
Chemokine receptors:
These GPCRs regulate immune cell migration and are central to inflammatory and autoimmune diseases. Biomarkers such as chemokine ligand levels or receptor expression patterns can stratify patients and predict responsiveness to therapies targeting immune pathways.
5 HT2A receptors:
Serotonin receptors, particularly 5 HT2A, are critical in psychiatric and neurological disorders. PET imaging and receptor occupancy biomarkers have been used to guide dose selection in antipsychotic and psychedelic derived therapies, ensuring both efficacy and safety.
Oxytocin receptors:
Oxytocin signaling influences social behavior, stress, and reproductive physiology. Biomarkers such as plasma oxytocin levels or receptor polymorphisms are being explored to predict treatment outcomes in conditions ranging from autism spectrum disorders to postpartum depression.
Neurokinin receptors:
These receptors play a role in pain perception and stress responses. Biomarkers such as substance P levels or receptor occupancy assays have been used to evaluate novel analgesics and antidepressants, helping determine whether drugs truly engage their intended targets.
Glucagon like peptide 1 (GLP 1) receptor:
Widely known for its role in metabolic regulation, the GLP 1 receptor is a major target in diabetes and obesity. Biomarkers such as blood glucose, insulin levels, and weight reduction serve as direct pharmacodynamic readouts, linking receptor activity to clinical benefit.
Anti GPCR autoantibodies:
In certain autoimmune conditions, patients develop antibodies against GPCRs. These autoantibodies themselves can serve as biomarkers, both for diagnosis and for monitoring disease progression, offering new avenues for precision medicine in immunology.
GPCR PET tracers:
Positron emission tomography (PET) tracers provide non invasive biomarkers of GPCR activity in humans. They enable visualization of receptor binding and occupancy, offering direct evidence of target engagement and supporting dose optimization in clinical trials.
Together, these examples show how biomarkers can transform GPCR drug discovery from a high-risk endeavor into a more predictable process.
Biomarkers in Translational Pharmacology
Biomarkers are not only useful in isolated case studies, they are fundamental to translational pharmacology itself. Their role is to bridge the gap between preclinical models and clinical application, ensuring that findings in cell lines or animal studies translate meaningfully into human outcomes. By confirming target engagement, monitoring pharmacodynamic effects, and stratifying patients, biomarkers provide the evidence needed to move discoveries “from bench to bedside.” Importantly, they also enable feedback loops: insights gained in the clinic can refine preclinical models, creating a cycle of continuous improvement. This bidirectional flow is what makes biomarkers indispensable to translational pharmacology and central to reducing attrition in drug development.
The Role of Artificial Intelligence and Precision Medicine
Biomarker discovery is evolving rapidly. Traditional laboratory assays remain important, but artificial intelligence (AI) and machine learning are now accelerating the process. These technologies can:
- Mine complex omics datasets to identify hidden biomarker signatures.
- Stratify heterogeneous patient populations, such as those with Alzheimer’s disease.
- Support adaptive trial designs that adjust in real time based on biomarker readouts.
This convergence of translational pharmacology, biomarkers, and AI is driving the shift toward precision medicine. Instead of one-size-fits-all therapies, treatments are increasingly tailored to individual biomarker profiles. For industry stakeholders, this means not only better outcomes for patients but also more efficient use of resources and higher chances of commercial success.
Implications for Pharma and Biotech
For pharma and biotech professionals, the message is clear: biomarkers are no longer optional. They are central to translational pharmacology, particularly in GPCR drug discovery. Their integration into development programs enhances efficiency, reduces attrition, and supports regulatory approval.
Strategically, biomarkers also create competitive advantage. Companies that invest in biomarker discovery and validation can design smarter trials, secure faster approvals, and differentiate their therapies in crowded markets. As regulators increasingly emphasize biomarker-driven evidence, early adoption becomes not just beneficial but essential.
This chapter provides a comprehensive overview of biomarker categories, GPCR case studies, and emerging technologies. It offers a roadmap for organizations seeking to de-risk development and accelerate innovation in a competitive landscape.
Conclusion
Biomarkers are redefining the trajectory of GPCR drug discovery. By aligning translational pharmacology with precision medicine and AI-driven discovery, the industry can move closer to delivering therapies that are both effective and individualized.
For pharma and biotech professionals, the takeaway is straightforward: embrace biomarkers early, integrate them strategically, and leverage them as a foundation for innovation. Doing so will not only improve scientific outcomes but also strengthen the business case for new therapies.
For a deeper exploration of biomarkers and their application in translational GPCR biology and beyond, see the book chapter Translational Pharmacology and Biomarkers Applied to GPCR.
Reference
Özcelik, D.; Andreetta, F.; van der Kam, E. L. (in press). Translational pharmacology and biomarkers applied to GPCR. Reference Module in Chemistry, Molecular Sciences and Chemical Engineering (planned for publication in the Comprehensive Medicinal Chemistry, 4e, edited by Rotella, D.; Ward, S.) Elsevier, 2026 doi: https://doi.org/10.1016/B978-0-443-29808-0.00036-4.